Home /
Genome editing to dissect genetic risk factors for osteoarthritis
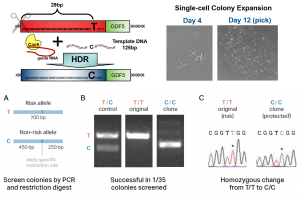
Genome-wide association studies (GWAS) have identified more than 100 genomic locations that harbor risk factors for the development of osteoarthritis (OA). However, almost all of the putative risk variants are located in non-coding areas of the genome and therefore it has been challenging to translate findings from GWAS into improved understanding and treatment of OA. We have developed a genome-editing workflow to introduce specific alterations in primary human chondrocytes. Single-cell derived colonies after editing are genetically defined and maintain the capacity to produce engineered cartilage and respond to catabolic stimuli. In collaboration with genomics collaborators, we are using this genome-editing approach to pinpoint the causal variants and target genes that mediate OA risk.
Further, known human genetic variants can be modeled in novel genetically-engineered mouse models as a way to study the mechanisms by which these subtle variations contribute to OA risk. For example, harboring two copies of an 8 base pair insertion in Chondroadherin-like (Chadl) is associated with a nearly 8-fold risk for total hip replacement. We have generated an "8 bp mouse" to understand how alterations to this extra-cellular matrix protein affect the properties of cartilage, and whether this variant has effects that are distinct from "just a loss of function".
Papers:
Thulson E, Davis ES, D’Costa S, Coryell P, Kramer NE, Mohlke KL, Loeser RF, Diekman BO&, Phanstiel DH&. 3D chromatin structure in chondrocytes identifies putative osteoarthritis risk genes. Genetics. 2022 Sep 13;iyac141. & = co-corresponding.
Richard D, Capellini TD, Diekman BO. Epigenetics as a mediator of genetic risk in osteoarthritis: role during development, homeostasis, aging, and disease progression. Am J Physiol Cell Physiol. 2023 May 1;324(5):C10178-C1088.
D’Costa S, Rich MJ, Diekman BO. Engineered cartilage from human chondrocytes with homozygous knockout of cell cycle inhibitor p21. Tissue Engineering Part A. 2019 Nov 22. doi: 10.1089/ten.TEA.2019.0214.
Funding:
NIH R21, 1R21AR077821-01A1
NC TraCS, $50k Pilot #550KR272106